Human African trypanosomiasis, also known as sleeping sickness, is a vector-borne parasitic disease. Its causative agents are parasitic protozoa belonging to the genus Trypanosoma. They are transmitted to humans by the bite of a tsetse fly (genus Glossina), infected from humans or animals that act as hosts of these parasites that are pathogenic to humans.
The tsetse fly is found only in sub-Saharan Africa, and only a few species can transmit the disease. For currently unknown reasons, cases of sleeping sickness are not reported in many areas of the tsetse fly's habitat. Residents of endemic rural areas engaged in agriculture, fishing, livestock raising and hunting are most susceptible to tsetse fly bites and, consequently, to this disease. Cases of the disease can occur both in individual villages and in the entire region. Within a single infected area, the intensity of the spread of the disease can vary greatly from one rural settlement to another.
Forms of human African trypanosomiasis
Human African trypanosomiasis has two forms depending on the type of pathogen:
- Trypanosoma brucei gambiense is found in 24 countries in West and Central Africa. Infection with this type of parasite (currently 95% of all reported cases of sleeping sickness) results in chronic infection. A person can carry the infection for many months or even years without showing any serious signs or symptoms of the disease. Often more severe symptoms occur at a later stage of the disease, when the central nervous system is affected.
- Trypanosoma brucei rhodesiense is found in 13 countries in Eastern and Southern Africa. Infection with this type of parasite (currently accounting for less than 5% of reported cases of sleeping sickness) causes an acute infection. The first signs and symptoms appear several months or weeks after infection. The disease develops rapidly with damage to the central nervous system. Uganda is the only country where both forms of the disease occur, although their distribution areas do not coincide.
There is another form of trypanosomiasis that occurs primarily in Latin America. It is known as American trypanosomiasis or Chagas disease. The causative agent of Chagas disease belongs to a different subgenus of Trypanosoma, is transmitted by other vectors and causes a disease whose clinical manifestations differ from human African trypanosomiasis.
Types of tsetse flies
It’s hard to imagine how much effort and probably suffering it took for entomologists to study and group dangerous insects into separate groups:
- representatives of the morsitans group live mainly in savannah forests and savannas, easily adapting to a relatively dry climate. It was these flies that contributed to the expansion of the area of wooded savannas, which is why Sierra Leonean pastoralists stopped raising horses;
- species of the fusca group are typical inhabitants of tropical rainforests; their bites are mainly inflicted on wild mammals, and less often on people and livestock;
- Tsetse flies from the palpalis group are the most moisture-loving, but they are attracted not by jungle swamps, but by open areas along river banks, where fishermen and swimmers most often become victims of insects.
It is in such natural conditions that the most dangerous insects on the planet live and breed safely, because they do not experience a shortage of food sources.
Animal trypanosomiasis
Other species and subspecies of parasites of the genus Trypanosoma are pathogenic for animals and cause trypanosomiasis in some species of wild and domestic animals. Trypanosomiasis in cattle is called Nagana. Trypanosomiasis in domestic animals, especially cattle, seriously undermines the economic development of infected rural areas.
Animals can harbor parasites pathogenic to humans, especially Tb rhodesiense, with domestic and wild animals being important reservoirs for the latter. Animals can also be infected with Tb gambiense and may also act as reservoirs, although to a lesser extent. However, the actual role of the animal reservoir in the epidemiology of the Gambian form of the disease is still not well understood.
Distinctive features
The insect is not characterized by the presence of poisonous glands and, in general, looks quite harmless. Its miniature size does not cause visual alarm, but you should remember the structure of this creature in order to avoid unpleasant encounters. The special position of the wings above the body indicates that the fly belongs to this species. She puts them one on top of the other. And if you look closely, you will notice a characteristic plumage pattern that is similar to the shape of an ax.
In addition, a distinctive feature is a peculiar proboscis capable of injecting. Plus, this insect is given away by its size; the length of the fly reaches from 9 to 14 mm. This variety can be recognized by its reddish-gray chest, which is divided by 4 reddish stripes along the body. It is also distinguished by the color of its abdomen, which is yellow on top and gray underneath. Adding to the individual characteristics are hair antennae growing on the insect's head.
Major epidemics
Several epidemics have occurred in Africa over the past century:
- the epidemic of 1896-1906, mainly affecting Uganda and the Congo River basin;
- the 1920 epidemic that affected a number of African countries; And
- the most recent epidemic, which began in 1970 and continued until the end of the 1990s.
In 1920, the epidemic was fought with mobile teams that screened millions of people living in high-risk areas. By the mid-1960s. the spread of the disease was brought under control, with fewer than 5,000 cases reported throughout the continent. Following this success, surveillance was relaxed, causing the disease to return, and by 1970, outbreaks had grown to epidemic levels in several regions. In the 1990s. and at the beginning of the 21st century, thanks to the efforts of WHO, the implementation of national programs to combat the disease, bilateral cooperation and the activities of non-governmental organizations (NGOs), the upward trend in incidence was stopped.
As the number of new cases of human African trypanosomiasis decreased significantly between 2000 and 2012 as a result of international coordinated efforts, the WHO Roadmap for Control of Neglected Tropical Diseases set a goal for its elimination as a public health problem by 2022 and elimination transmission (reducing the number of cases to zero) by 2030.
Fighting methods
The first attempts to get rid of the tsetse fly were made at the beginning of the last century, then hundreds of thousands of animals were exterminated: elephants, lions and ungulates. However, this did not affect the state of the tsetse population in any way; the flies survived by feeding on the blood of mouse-like rodents, birds and amphibians, which are also carriers of trypanosomiasis.
Cutting down the bushes also did not bring success, and in the 40s the main method of control was the use of the insecticide DDT against insects. But this turned out to be a temporary measure: like most insect pests, the tsetse’s body quickly acquires immunity to pesticides.
Not all tsetse fly species are carriers of trypanosomiasis, but visitors to exotic Africa who are bitten by an insect with characteristic symptoms are advised to seek immediate medical attention.
Burden of disease
Sleeping sickness threatens the health of millions of people in 36 countries in sub-Saharan Africa. Many of them live in remote areas with limited access to necessary medical care, making surveillance and therefore difficult to diagnose and treat patients. In addition, population displacement, armed conflict and poverty are important factors facilitating transmission.
- In 1998, there were almost 40,000 cases of the disease, but the estimated number of undiagnosed patients who did not receive treatment was about 300,000.
- During the most recent epidemic, disease prevalence reached 50% in some rural communities in Angola, the Democratic Republic of the Congo and South Sudan. In these communities, sleeping sickness was the first or second leading cause of death, surpassing even HIV/AIDS.
- In 2009, systematic efforts to control the disease brought the number of reported cases to below 10,000 (9,878 cases) for the first time in 50 years. The decline in incidence continued in subsequent years, with 992 new cases reported in 2022, the lowest number in 80 years of systematic global collection of incidence data. The population at risk of the disease is estimated to be 65 million.
Current distribution of the disease
Prevalence rates of the disease vary between countries, as well as between areas within a given country.
- Over the past 10 years, more than 70% of cases have been reported in the Democratic Republic of the Congo.
- Angola, Cameroon, Central African Republic, Chad, Congo, Guinea, Malawi, South Sudan and Zambia reported between 10 and 100 new cases in 2022, while Côte d'Ivoire, Equatorial Guinea, Gabon, Uganda, United The Republic of Tanzania and Zimbabwe – from 1 to 10 new cases.
- Over the past 10 years, isolated cases have been reported in countries such as Burkina Faso, Ghana and Nigeria.
- Benin, Botswana, Burundi, Ethiopia, Gambia, Guinea-Bissau, Liberia, Mali, Mozambique, Namibia, Niger, Rwanda, Senegal, Sierra Leone, Eswatini and Toga have reported no new cases in the past 10 years. In some of these countries, transmission appears to have stopped, but it is difficult to accurately assess the real situation in some areas because surveillance and diagnosis are hampered by social instability and/or difficulties in accessing the population.
Habitats
The tsetse fly is widespread in Africa. The habitat of obligate parasites includes countries located south of the Sahara Desert.
The tsetse fly lives in 37 countries, most of which are the poorest in the world. Due to the extremely low social level of the states, the authorities are unable to use chemicals against parasites and provide the population with medications that could save the population from death.
Tsetse flies, which carry a dangerous disease, are common in areas of the African continent that are free from people and have the status of wildlife reserves.
It should be noted that it is the peculiarities of the habitat of insects dangerous to humans that prevent people from developing fertile land.
Video
WORLD OF INSECTS - FLIES: Tsetse FLY
Mechanism of transmission and symptoms
The disease is transmitted mainly by the bite of an infected tsetse fly, but other mechanisms of transmission are known:
- mother-to-child transmission: trypanosomes can cross the placenta and infect the fetus;
- mechanical transmission through other blood-sucking insects is possible, but the epidemiological significance of this route of transmission is unknown;
- There are known cases of accidental infection of laboratory personnel as a result of injury from a contaminated needle;
- There is information about the transmission of the parasite through sexual contact.
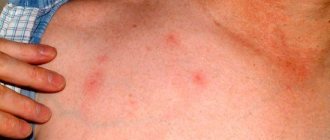
During the first stage of infection, trypanosomes multiply in subcutaneous tissues, blood and lymph. This stage is known as the hemolymphatic phase of the disease, which is characterized by bouts of fever, headache, swollen lymph nodes, joint pain and itching.
In the second stage, the parasites cross the blood-brain barrier and infect the central nervous system. This stage is known as neurological, or meningoencephalitic. In general, the most obvious signs and symptoms of the disease appear at this stage: behavioral changes, confusion, sensory disturbances and loss of coordination. An important clinical sign at this stage is a disturbance in the sleep cycle, which gives the disease its name. Without treatment, sleeping sickness is considered fatal, although there are known cases of healthy carriers of the infection.
What does the tsetse fly eat?
The tsetse fly is a typical representative of obligate hematophages. The diet of insects in the adult stage consists exclusively of animal and human blood, which they obtain through the special structure of their oral apparatus.
Tsetse fly, mouthparts visible.
The proboscis of the tsetse fly is of a cutting-sucking type, perfectly adapted for gnawing the skin of such large animals as a warthog, buffalo, antelope or horse. What can we say about human skin?
Tsetse fly before lunch.
When a tsetse fly bites, it secretes saliva and a blood-thinning enzyme into the wound, after which it becomes saturated with blood, and its abdomen doubles in size.
Like most relatives, tsetse flies do not suffer from a lack of appetite, but surprisingly, they cannot boast of the same fertility.
Disease management: making a diagnosis
Disease management is carried out in three stages:
- Screening for possible infection. This includes serological testing (only available for Tb gambiense) and testing for clinical signs, particularly enlarged cervical lymph nodes.
- Search for parasites in biological fluids.
- Determining the stage of the disease. This involves a clinical examination of the patient and, in some cases, examination of cerebrospinal fluid obtained by lumbar puncture.
To avoid the need for complex and difficult treatment methods, diagnosis should be performed as early as possible before the development of the neurological stage of the disease.
Due to the long and asymptomatic course of the first stage of sleeping sickness caused by T. b. gambiense, careful and active screening of risk groups is recommended in order to identify patients at an early stage of the disease and limit further spread of infection through their treatment, after which they cease to act as a reservoir. Comprehensive population screening requires significant financial investment in human and material resources. In Africa, especially in remote areas where the disease is most prevalent, these resources are often in short supply. As a result, some infected people die before being diagnosed and receiving treatment.
Treatment
The type of treatment depends on the form and stage of the disease. Early detection of the disease increases the chances of successful treatment. Because the parasite can remain viable for a long time and cause relapses many months after the end of therapy, patients should remain under observation for up to 24 months to evaluate the results of treatment, during which clinical examination of patients and laboratory tests should be performed. examination of bodily fluids, including, in some cases, cerebrospinal fluid obtained by lumbar puncture.
To successfully treat the disease at the second stage, it is necessary to use drugs that can overcome the blood-brain barrier and neutralize the parasite in the central nervous system.
In 2022, WHO published new guidelines for the treatment of the Gambian form of human African trypanosomiasis. A total of six drugs are registered for the treatment of sleeping sickness. These drugs are provided to WHO by manufacturers free of charge and supplied free of charge to endemic countries.
Drugs for the treatment of the disease at the first stage:
- Pentamidine: first developed in 1940, used to treat sleeping sickness caused by Tb gambiense in the first stage. Despite the presence of serious side effects, the drug is generally well tolerated.
- Suramin: first obtained in 1920, used to treat Tb rhodesiense disease in the first stage. The drug has a number of undesirable side effects, including nephrotoxicity and allergic reactions.
Drugs for the treatment of the disease in the second stage:
- Melarsoprol: first introduced in 1949, used to treat infections caused by both pathogens. It is an arsenic derivative and causes numerous unwanted side effects, the most severe of which is reactive encephalopathy (encephalopathic syndrome), which can be fatal (in 3%–10% of cases). It is currently recommended as a first-line treatment for the Rhodesian form of the disease, but is rarely prescribed for the Gambian form of infection.
- Eflornithine: registered in 1990, much less toxic than melasoprol, effective only against Tb gambiense. It is usually used in combination with nifurtimox (as part of combination nifurtimox-eflornithine therapy), although it can also be used as monotherapy. The treatment regimen is complex and inconvenient.
- Nifurtimox: Nifurtimox-eflornithine combination therapy was proposed in 2009. It simplifies the use of eflornithine by reducing the duration of treatment and the number of intravenous perfusions, but unfortunately its effectiveness against Tb rhodesiense has not yet been studied. Nifurtimox is registered for the treatment of American but not African human trypanosomiasis. WHO supplies both drugs to endemic countries free of charge, complete with all consumables necessary for their use.
Drugs to treat the disease at both stages:
Fexinidazole is an oral drug for the treatment of the Gambian form of human African trypanosomiasis. In 2022, the drug was included in the WHO List of Essential Medicines and recommended in WHO guidelines for the treatment of human African trypanomosis. The drug is indicated as first-line therapy in the first and mild second stages of the disease. Treatment should be carried out under the supervision of qualified medical personnel; The drug is taken 30 minutes after eating solid food. Clinical trials of this drug are currently underway for the treatment of the Rhodesian form of trepanosomiasis.
Public-private partnerships
In 2000 and 2001 WHO established a public-private partnership with Aventis Pharma (now Sanofi) and Bayer HealthCare, resulting in a WHO-led disease surveillance and control program that provides control support to endemic countries at no cost to medicines are supplied.
The partnership has been renewed several times in 2006, 2011, 2016 and 2021. Progress in reducing the incidence of sleeping sickness and the prospect of its elimination has prompted a number of other private sector partners to support the WHO initiative to eliminate the disease as a public health problem and ensure the elimination of transmission by 2030 G.
How to get rid of tsetse flies?
As already mentioned, scientists are trying to find a way to destroy insects, but so far they have not succeeded.
To combat the tsetse fly in the 20th century, the following methods were used:
- Scorched earth. To do this, they killed all the livestock that the tsetse fly fed on. Because of this, the insects died. But this method was effective only in the short term; in the long term it turned out to be useless, because the fly fed not only on large animals, but also on reptiles and rodents.
- Deforestation. This method also cannot be called effective. As the name suggests, it involved cutting down forests so that the tsetse fly would lose its usual living conditions. But it is obvious that this method only brings harm, not benefit.
- Use of pesticides. To do this, special preparations were sprayed from airplanes and applied to animals. But the method did not bring the desired effect.
- Setting traps. A simple mechanical method gives good results. To make a trap, you need to take dark cloth or buffalo hide and soak it with insecticides. To attract flies, an artificially created smell is used, reminiscent of an animal's breath or urine. This method helped reduce the tsetse fly population.
- Sterilization of males. At the moment, the method is the most effective. To sterilize the males, they were irradiated with radiation and released into places where insects were crowded. After mating, the females were unable to produce offspring, and their eggs remained unfertilized. This method was especially effective in Zanzibar. Its disadvantage is the lack of a water barrier with neighboring states, as a result of which healthy males entered the region, and tsetse flies multiplied again.
The tsetse fly is a terrible insect that causes serious harm to animals and people. But at the same time, there are people who believe that the insect is necessary. In particular, wildlife conservationist Bernhard Grzimek believes that the tsetse fly protects pristine nature from the invasion of civilization.